You are losing hair. You are gaining weight around your abdomen. You feel anxious in the second half of your cycle in ways that do not match your circumstances. Your sleep is poor in the week before your period. You feel flat, unmotivated, and emotionally reactive.
These look like separate problems with separate causes. They are not. They are the predictable downstream effects of one hormone running consistently below the level your body requires to maintain the systems that depend on it.
Progesterone does not just govern the menstrual cycle. It governs mood regulation through its conversion to neurosteroids. It governs body composition through its opposition of oestrogen. It governs hair follicle cycling, sleep architecture through its interaction with GABA, and the basic sense of calm that a healthy hormonal environment produces. When it drops, everything it maintains degrades simultaneously. This post explains the mechanism behind that drop and what the research says about correcting it.
Do You Recognise This Pattern?
Before the mechanism, a brief check. Mark how many of these apply to you consistently, particularly in the second half of your cycle.
Your hair is thinning or shedding more than it used to, diffusely across the scalp rather than in a defined pattern.
You are gaining weight particularly around the abdomen and it is resistant to the changes in diet or exercise that would normally produce results.
Your mood shifts noticeably in the week or two before your period: anxiety, irritability, low motivation, or a flat emotional quality that lifts when your period arrives.
Your sleep is worse in the second half of your cycle, particularly difficulty staying asleep or waking early.
Your PMS has worsened over the past one to two years without a clear dietary or lifestyle explanation.
If you are in perimenopause: your cycles are becoming irregular, the symptoms above are intensifying, and the fluctuations feel like they are accelerating.
If you recognise four or more of these, particularly with the cyclical timing pattern, low progesterone is a strong candidate for the common driver. The mechanism below explains why one hormone produces this range of effects simultaneously.
BEFORE YOU READ FURTHER This post describes one specific and well-documented mechanism behind the symptoms above: progesterone deficiency driven primarily by the pregnenolone steal from chronic cortisol elevation, compounded by mineral depletion of the enzymatic cofactors that produce progesterone. This is a real and common mechanism in otherwise healthy women. It is not the only mechanism. If you are pregnant and experiencing low progesterone symptoms, this is a clinical matter requiring immediate guidance from your midwife or obstetrician. This post is not the appropriate resource for that situation. Also see a GP if symptoms are severe, appeared suddenly, are accompanied by significant cycle irregularity, or if you have fertility concerns. These warrant full hormonal panel assessment before any supplementation approach. The mechanism described in this post is most relevant for women experiencing a gradual, worsening, cyclically-timed symptom pattern without a diagnosed underlying condition. If that is your situation, read on. |
What Actually Causes Low Progesterone
The standard answer is stress, age, or diet. These are true but they are surface descriptions. The biological mechanism underneath all three of them is the same, and understanding it is what tells you what to actually do about it.
The Pregnenolone Steal
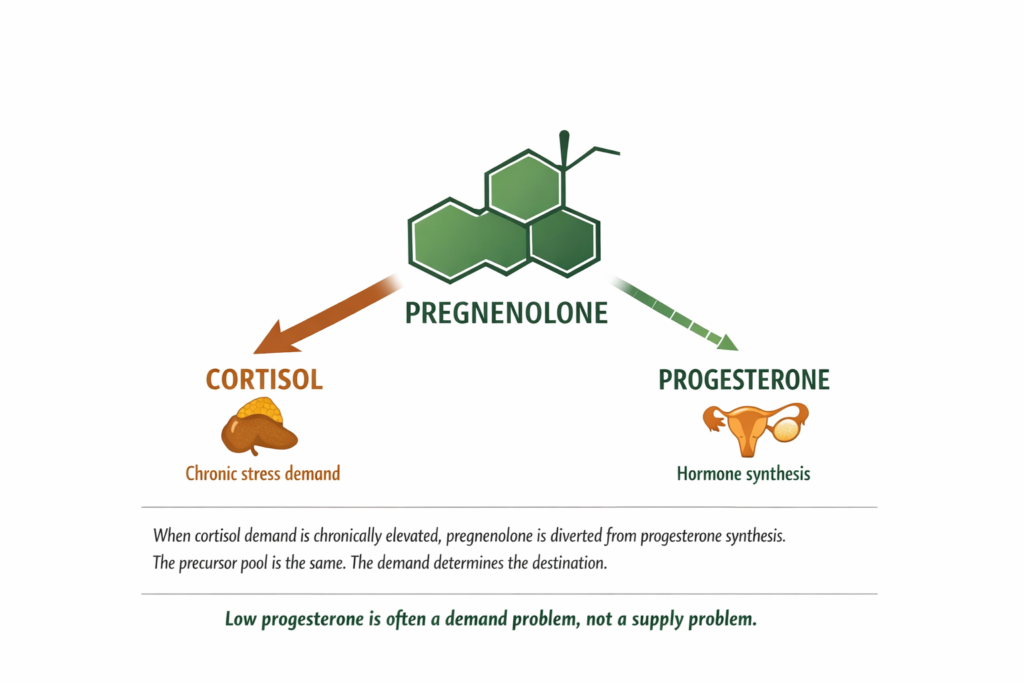
Progesterone is not produced directly. It is synthesised from a precursor molecule called pregnenolone. Pregnenolone is also the precursor for cortisol, testosterone, and the oestrogens. All of these hormones draw from the same upstream pool.
Under normal conditions the body distributes pregnenolone across multiple synthesis pathways in proportions that maintain hormonal balance. Under chronic stress, the HPA axis continuously signals the adrenal glands to produce cortisol. That demand is prioritised. The enzyme pathway toward cortisol synthesis receives more pregnenolone. The pathway toward progesterone receives less. Not because progesterone production is broken. Because the upstream substrate is being diverted.
This mechanism does not require a diagnosed stress disorder. It requires sustained cortisol activation over weeks or months. The person does not need to feel overwhelmed. They need only to have been under continuous low-grade activation, the kind produced by chronic work pressure, financial stress, or any situation that keeps the stress response from fully standing down between demands (Majewska et al., 1986; Epel et al., 2000).
The full cortisol mechanism, including how it depletes minerals and disrupts multiple hormone pathways simultaneously, is covered in our post on high cortisol symptoms and what the mineral depletion cycle actually does to your body.
DEFINITION PREGNENOLONE STEAL Pregnenolone is the master precursor molecule from which the body synthesises multiple steroid hormones including cortisol, progesterone, testosterone, and the oestrogens. The pregnenolone steal describes the condition in which chronic cortisol demand diverts pregnenolone preferentially toward cortisol synthesis, leaving less available for progesterone and other hormone pathways. The result is a relative progesterone deficiency even when no structural problem exists with the ovaries or adrenal glands. This mechanism is documented in the steroidogenesis literature and does not require a diagnosed adrenal condition to be clinically relevant. |
The Mineral Depletion Component
The conversion of pregnenolone to progesterone is catalysed by the enzyme 3-beta-hydroxysteroid dehydrogenase. This enzyme requires magnesium as a cofactor (Schierbauer et al., 2023). When magnesium is depleted, the conversion is impaired regardless of how much pregnenolone is available. The substrate is present. The tool required to convert it is not.
Zinc supports luteinising hormone production from the pituitary. Luteinising hormone is the signal that tells the corpus luteum after ovulation to produce progesterone in the luteal phase. When zinc is depleted, the luteinising hormone signal weakens. The corpus luteum receives a reduced signal and produces less progesterone (Netter et al., 1981). This is the mechanism behind luteal phase deficiency: not a structural problem with the corpus luteum but an insufficient signal from a mineral-depleted pituitary.
Cortisol depletes both magnesium and zinc with every activation. The same mechanism that diverts pregnenolone also removes the enzymatic tools required to convert whatever pregnenolone remains. The cortisol-mineral connection is not a single pathway. It is a double depletion of both the precursor and the cofactors simultaneously.
WHAT TO DO ABOUT IT The pregnenolone steal is a demand problem, not a supply problem. Adding progesterone precursors through food does not solve a demand problem. Reducing the cortisol demand that is causing the diversion does. Ashwagandha withanolides act directly on the glucocorticoid receptors in the hypothalamus that govern HPA axis feedback sensitivity. When those receptors regain sensitivity, the cortisol signal normalises. Pregnenolone is no longer diverted at the same rate. Progesterone synthesis resumes from the same precursor pool that was always there. Simultaneously, restoring ionic magnesium and zinc addresses the enzymatic cofactor depletion at the conversion step directly. |
DOES THIS APPLY TO YOU? If your symptoms have worsened or appeared during or following a period of sustained stress rather than emerging from nowhere, the pregnenolone steal mechanism is almost certainly active. If your symptoms are consistent across your entire cycle rather than worsening in the second half specifically, or if they appeared alongside a specific medical diagnosis or medication change, other mechanisms warrant investigation alongside this one. |
Understanding why the deficiency develops changes the intervention entirely. The same mechanism that explains the cause of the problem also reveals which of the following symptoms to expect and why they all trace back to a single origin.
Hair Loss: The Most Distressing Visible Symptom
Progesterone maintains hair follicle cycling by balancing the effects of androgens at the follicle level. Progesterone competes with dihydrotestosterone, a potent androgen, at the androgen receptors in hair follicles. When progesterone drops, dihydrotestosterone wins more receptor competition. The follicles shift from the growth phase toward the resting and shedding phase more readily and for longer. The result is diffuse thinning across the scalp rather than the defined recession pattern associated with androgenic alopecia (Prior, 2011).
The mineral depletion component operates simultaneously. Zinc is required for keratinocyte production, the cells that form the hair shaft. Low progesterone and low zinc therefore produce hair loss through two different mechanisms at the same time, which explains why the pattern can be so persistent and why addressing only one variable often produces incomplete results.
The cyclical timing is the diagnostic signal. If hair shedding is noticeably worse in the two weeks before your period and slightly improved after it begins, the progesterone-androgen receptor mechanism at the follicle level is the most likely driver. Diffuse pattern across the whole scalp rather than the temples or hairline confirms this over androgenic causes.
WHAT TO DO ABOUT IT Restoring progesterone through the cortisol pathway normalises the androgen receptor balance at the follicle. Restoring ionic zinc addresses the keratinocyte production step directly. Because the hair follicle growth cycle takes three to six months to complete, meaningful improvement in hair density typically takes that long even when the hormonal environment has already normalised. The absence of change in the first four to six weeks is not evidence that the mechanism has not been addressed. It is evidence of the biological timeline of follicle cycling. |
DOES THIS APPLY TO YOU? If your hair loss is diffuse rather than patterned, has worsened alongside other cyclical symptoms, and coincides with a period of sustained stress, the progesterone-androgen receptor mechanism fits. If loss is in a defined pattern at the hairline or temples, appeared suddenly, or is accompanied by scalp changes, a GP assessment to rule out thyroid, autoimmune, or androgenic alopecia causes is the appropriate first step before treating this as a progesterone issue. |
Hair loss and weight gain are the two symptoms that draw the most search attention in this cluster. They are also the two symptoms that respond most clearly once the hormonal environment has been corrected. The weight gain mechanism is different from the hair loss mechanism but traces to the same upstream cause.
Abdominal Weight Gain: Why the Abdomen Specifically
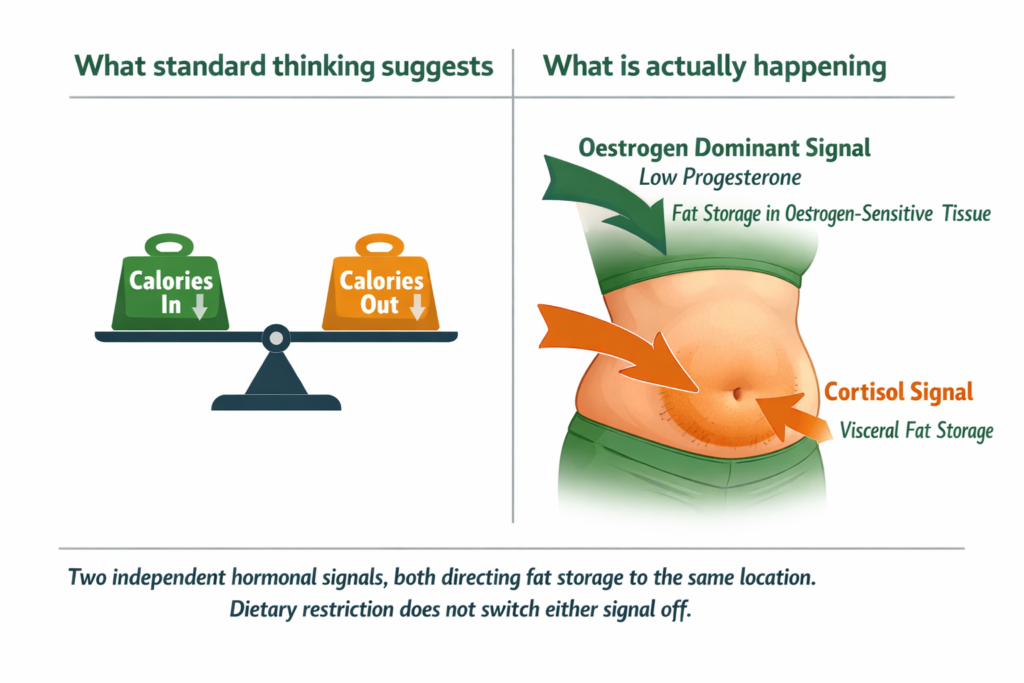
Progesterone and oestrogen are hormonal opposites in several of their effects on body composition. Progesterone opposes oestrogen’s action on fat storage, particularly in the abdominal region. When progesterone drops without a corresponding drop in oestrogen, the balance shifts toward relative oestrogen dominance. Oestrogen in excess of progesterone promotes fat storage in oestrogen-sensitive tissue, which in adult women is concentrated in the abdomen, hips, and thighs.
The cortisol component compounds this directly. Cortisol independently stimulates abdominal fat storage through glucocorticoid receptors concentrated in visceral fat tissue. The person with low progesterone from the pregnenolone steal is simultaneously experiencing elevated cortisol and its independent fat storage effect. Two separate hormonal mechanisms are pointing at the same location.
DEFINITION OESTROGEN DOMINANCE A state in which the ratio of oestrogen to progesterone is elevated relative to optimal hormonal balance. Does not require total oestrogen to be high. Can occur when progesterone falls while oestrogen remains stable or declines more slowly. The resulting symptom cluster includes abdominal weight gain, breast tenderness, heavy or irregular periods, water retention, and mood instability. The pregnenolone steal is one of the most common mechanisms producing relative oestrogen dominance in otherwise healthy women in their 30s and 40s. |
WHAT TO DO ABOUT IT Dietary restriction alone does not address this because the hormonal environment driving the fat storage remains active regardless of caloric intake. The intervention is hormonal, not caloric. Restoring progesterone through the cortisol pathway changes the oestrogen-progesterone ratio. When progesterone rises, the oestrogen-dominant fat storage signal weakens. The abdominal accumulation responds over eight to twelve weeks of hormonal normalisation, not over days of restriction. |
DOES THIS APPLY TO YOU? If weight is accumulating specifically around your abdomen and is resistant to changes in diet or exercise that would normally produce results, and if it appeared or worsened during a period of sustained stress or hormonal transition, the progesterone-oestrogen imbalance mechanism fits. If weight gain is evenly distributed or directly proportional to changes in food intake, the hormonal mechanism is less likely to be the primary driver and caloric balance or thyroid function warrants investigation. |
The abdominal weight and the hair loss are the most visible expressions of the same underlying imbalance. The less visible symptoms, the ones that affect how you feel rather than how you look, operate through a third mechanism that is arguably more disruptive to daily life.
Mood, Sleep, and Fatigue: The Neurosteroid Connection
Progesterone is converted in the brain to a neurosteroid called allopregnanolone. Allopregnanolone is one of the most potent natural modulators of GABA receptor function known to neuroscience. GABA is the nervous system’s primary calming neurotransmitter. When progesterone is adequate, allopregnanolone keeps GABA receptor sensitivity in the range that produces emotional stability, restful sleep, and the capacity to return to calm after stress. When progesterone drops, allopregnanolone drops with it. GABA receptor function degrades (Backstrom et al., 2014).
The experience is biochemical anxiety that arrives without a clear situational trigger, particularly in the premenstrual phase. Sleep becomes fragmented in the second half of the night. Emotional reactivity increases out of proportion to circumstances. The flat, unmotivated quality that some women describe in the luteal phase reflects a nervous system running without adequate neurosteroid support, not a psychological state.
Fatigue in this pattern is partly neurological and partly energetic. Low progesterone impairs sleep quality through the GABA mechanism, creating compounding tiredness. Low magnesium from the cortisol depletion cycle simultaneously impairs cellular energy production through its role in ATP synthesis and sodium-potassium pump function. The fatigue from both pathways arrives together.
The full mechanism of how magnesium restores GABA function and sleep architecture is covered in our post on why magnesium glycinate specifically improves sleep quality and the 3am waking pattern.
WHAT TO DO ABOUT IT Restoring progesterone through the cortisol pathway directly increases allopregnanolone production. Restoring ionic magnesium supports GABA receptor function from the mineral side simultaneously. These two approaches address the same downstream symptom from complementary upstream mechanisms. Most women notice mood and sleep changes within two to four weeks of consistent ionic mineral and adaptogenic support as GABA function improves, before the full hormonal correction completes. |
DOES THIS APPLY TO YOU? If mood instability, anxiety, and poor sleep are specifically worse in the ten to fourteen days before your period and noticeably better once it begins, the progesterone-allopregnanolone-GABA mechanism describes your pattern precisely. If symptoms are consistent throughout the month without cyclical variation, other mechanisms including thyroid function, clinical anxiety, or iron deficiency warrant investigation alongside this one. |
The cyclical mood and sleep disruption is one of the clearest diagnostic signals that progesterone rather than another hormonal imbalance is the primary driver. In perimenopause, this pattern intensifies for a specific structural reason that is worth understanding separately.
Perimenopause: Why Everything Accelerates
In perimenopause, progesterone declines earlier and more steeply than oestrogen. The hormonal fluctuations that characterise this transition are largely driven by this asymmetry: oestrogen fluctuates but maintains an average that is not yet dramatically reduced, while progesterone drops consistently. The result is an intensification of the relative oestrogen dominance pattern described above, now compounded by declining ovarian function rather than purely by the pregnenolone steal.
The pregnenolone steal remains relevant in perimenopause because the adrenal glands become an increasingly important site of steroid hormone production as ovarian function declines. If those adrenal glands are simultaneously being tasked with continuous cortisol production from chronic stress, the pregnenolone available for adrenal progesterone synthesis is again diverted. The perimenopausal woman under chronic stress is experiencing both the natural decline of ovarian progesterone and the additional cortisol-driven suppression of adrenal progesterone simultaneously.
Women in their late 30s and early 40s often describe their symptoms as PMS that never fully resolves, cycles that are becoming shorter or more irregular, and a general sense that their hormonal environment is less stable than it used to be. This is the perimenopause transition beginning, often five to ten years before the final menstrual period, and the pregnenolone steal accelerates it measurably in women under sustained stress.
WHAT TO DO ABOUT IT Supporting the adrenal pathway toward progesterone synthesis becomes more important in perimenopause, not less, precisely because the ovarian pathway is declining. Reducing cortisol demand through ashwagandha and restoring the mineral cofactors through ionic magnesium and zinc addresses what the adrenals can produce from the pregnenolone that remains available when cortisol demand is normalised. This does not replace the oestrogen and progesterone the ovaries are no longer producing in the same quantities. It supports the adrenal contribution more fully. |
DOES THIS APPLY TO YOU? If you are between 38 and 55 and your symptoms have intensified over the past one to two years, particularly if cycles are becoming irregular or shorter, the perimenopausal pattern is very likely active. Significant hormonal disruption during perimenopause warrants GP or gynaecologist assessment for the full hormonal picture, including oestrogen and progesterone levels measured at the correct point in your cycle, alongside any adaptogenic and mineral support approach. |
How to Increase Progesterone Naturally: What the Mechanism Actually Requires
The honest answer to how to increase progesterone naturally is more specific than most sources provide.
You cannot eat progesterone. No food contains meaningful quantities of progesterone in a form the body can use directly. The most important first question is: what is sustaining the cortisol elevation that is causing the pregnenolone diversion?
If the answer is a sustained life stressor, addressing that stressor is the primary intervention. Ashwagandha and ionic minerals support the biological pathway and accelerate recovery, but they cannot fully restore progesterone production while the cortisol demand driving the pregnenolone steal remains active. Reducing the stressor load first, then supporting the biology, produces better and more durable results than the biological support alone.
For women whose cortisol is elevated from genuine life circumstances rather than purely biochemical dysregulation, the mineral and adaptogenic approach works as a parallel support layer while the primary stressor is being addressed, not as a substitute for addressing it.
Reducing Cortisol Demand: The Most Significant Lever
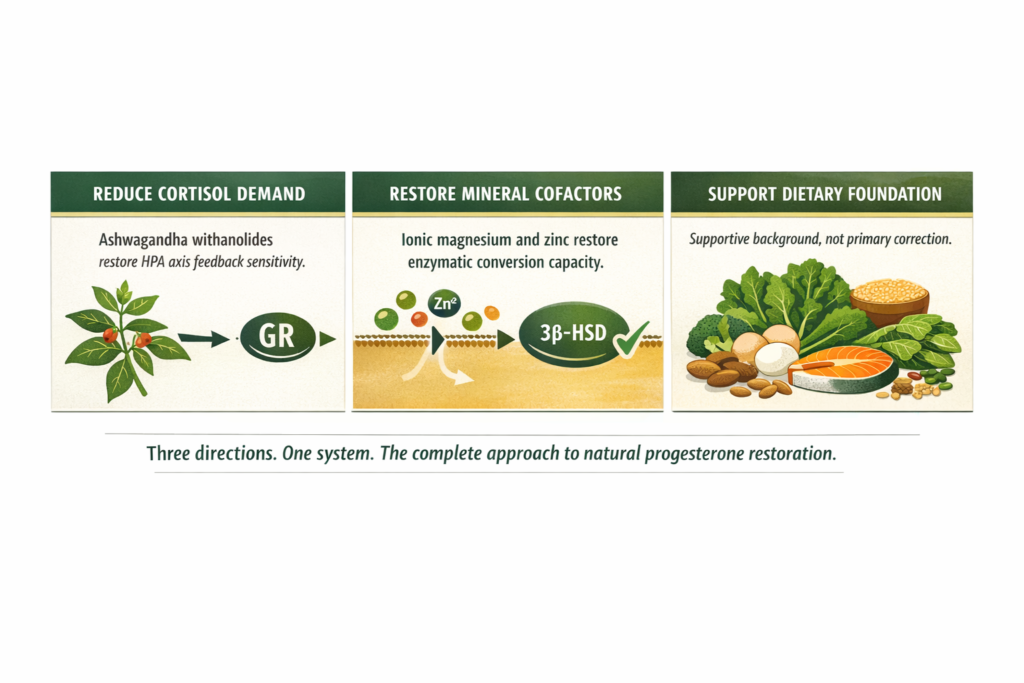
This requires both the lifestyle component, which is managing the sustained activation that drives chronic cortisol elevation, and the adaptogenic component. Ashwagandha does not simply calm you down. It restores the HPA axis feedback sensitivity that allows the stress response to stand down after activation has occurred. When that feedback sensitivity is restored through withanolide action on the glucocorticoid receptors, cortisol returns to its natural rhythm. Pregnenolone is no longer diverted at the same rate. The mechanism that was suppressing progesterone is addressed at its hormonal root rather than symptomatically.
Clinical research confirms this mechanism. A randomised controlled trial published in the Indian Journal of Psychological Medicine demonstrated significant reductions in serum cortisol in adults taking standardised ashwagandha extract versus placebo (Chandrasekhar et al., 2012). The mechanism is not a supplement claim. It is a documented receptor interaction with measured downstream hormonal effects.
The full explanation of how ashwagandha withanolides restore HPA axis feedback sensitivity is covered in our post on what adaptogens actually do at the hormonal level.
Restoring Mineral Cofactors: The Enzymatic Requirement
Ionic magnesium supports the 3-beta-hydroxysteroid dehydrogenase conversion step. Ionic zinc supports luteinising hormone production at the pituitary. Both need to be in their ionic form because standard supplement forms such as magnesium oxide and zinc oxide deliver a fraction of their stated dose to the intracellular level where enzymatic reactions occur. A 300mg capsule of magnesium oxide delivers approximately 12mg of usable magnesium to the cell. Ionic magnesium delivers 85 to 95 percent of its stated dose directly. The enzymatic cofactor requirement is intracellular. The form must match the location where the enzyme operates.
We cover the full framework for mineral form and bioavailability in our post on how mineral form determines what your cells actually receive.
The Food Contribution: What It Does and What It Cannot Do
Certain foods support the upstream conditions that favour progesterone production. Foods rich in zinc support the luteinising hormone signal: red meat, shellfish, pumpkin seeds. Foods rich in magnesium support the enzymatic conversion: dark leafy greens, dark chocolate, seeds, legumes. Foods rich in vitamin B6, a cofactor in steroid hormone synthesis: poultry, fish, potatoes, bananas. Foods rich in cholesterol, the starting molecule in all steroid hormone synthesis: eggs, quality dairy.
The honest limitation: soil mineral depletion since 1950 has reduced the zinc and magnesium content of these foods by 40 to 80 percent compared to historical nutritional data (Davis et al., 2004). Eating the right foods supports the direction of travel. For someone with established deficiency from a sustained cortisol depletion period, food sources alone are rarely sufficient to close the gap. They work alongside ionic mineral supplementation, not instead of it.
WHAT TO DO ABOUT IT The most complete approach works in three directions. First: identify and address the life circumstances or sustained stressors driving the cortisol elevation. This is the primary intervention. Second: reduce the HPA axis activation through ashwagandha withanolides acting on glucocorticoid receptor sensitivity, supporting the system’s capacity to stand down while the primary stressor is being addressed. Third: restore the enzymatic cofactors that the conversion pathway requires, ionic magnesium for the 3-beta-HSD conversion step and ionic zinc for the luteinising hormone signal. The biology recovers faster when the primary stressor is reducing and the biological support is in place simultaneously. |
DOES THIS APPLY TO YOU? If you have already made dietary changes toward zinc and magnesium-rich foods and noticed partial improvement without full resolution, the cortisol demand component is almost certainly still active. The food approach closes the supply side. The adaptogenic approach closes the demand side. Both are required for the mechanism to restore fully. If you have been taking magnesium supplements without result, verify the form on the label. Magnesium oxide at 4 percent absorption does not restore intracellular enzyme cofactor levels regardless of the stated dose. |
Standard Thinking vs. The Biological Reality
Standard Thinking | The Biological Reality |
|---|---|
Low progesterone is an oestrogen replacement problem | Low progesterone from the pregnenolone steal is a cortisol demand problem. Adding hormones without reducing the demand that causes the diversion addresses the symptom without the mechanism |
Eat more progesterone-boosting foods | No food contains bioavailable progesterone. Food supports the upstream enzymatic conditions but does not replace ionic mineral cofactors or address cortisol-driven pregnenolone diversion at the demand side |
Stress causes hormonal problems in a vague general sense | Cortisol specifically diverts pregnenolone from progesterone synthesis through a documented enzyme pathway competition. The mechanism is precise, biochemically verifiable, and not dependent on perceived stress level |
Low progesterone is just part of getting older | The natural decline of progesterone with age is real. Its acceleration through the pregnenolone steal from chronic stress and mineral depletion is an addressable mechanism, not an inevitable part of ageing |
Take magnesium and the conversion will improve | The form of magnesium determines whether it reaches the intracellular enzymatic level where the conversion occurs. Magnesium oxide at 4 percent absorption does not restore the cofactor at the enzyme site regardless of stated dose |
Frequently Asked Questions
What are the most common symptoms of low progesterone?
The most consistent symptoms cluster around cyclical timing: worsening anxiety and mood instability in the one to two weeks before menstruation, poor sleep in the luteal phase particularly in the second half of the night, diffuse hair thinning across the scalp, abdominal weight gain resistant to dietary change, and fatigue that improves after the period begins. In perimenopause these symptoms intensify as progesterone declines more steeply than oestrogen. The cyclical worsening and improvement pattern is the most reliable signal distinguishing low progesterone from other hormonal causes.
How do I increase progesterone naturally?
The most mechanistically grounded approach works in two directions simultaneously. First, reduce the cortisol demand that is diverting pregnenolone away from progesterone synthesis through adaptogenic support, specifically ashwagandha acting on glucocorticoid receptor sensitivity to restore HPA axis feedback. Second, restore the mineral cofactors that the progesterone synthesis enzymes require: ionic magnesium for the 3-beta-hydroxysteroid dehydrogenase conversion step and ionic zinc for the luteinising hormone signal from the pituitary. Dietary changes supporting these minerals provide useful background but are insufficient alone when modern soil depletion has reduced food mineral content by 40 to 80 percent since 1950.
Can stress cause low progesterone?
Yes, through a specific and documented mechanism called the pregnenolone steal. Chronic cortisol demand preferentially diverts the shared precursor pregnenolone toward cortisol synthesis and away from progesterone synthesis. Cortisol also depletes magnesium and zinc with each activation, removing the enzymatic cofactors that the progesterone synthesis pathway requires. The result is a dual suppression of progesterone: reduced precursor availability and impaired enzymatic conversion simultaneously. This explains why sustained stress, not just acute stress, is the relevant variable.
Why is my progesterone low even though I eat well?
Two reasons that operate independently of dietary choices. First, soil mineral depletion since 1950 has reduced the zinc and magnesium content of vegetables and wholegrains by 40 to 80 percent compared to historical data. Foods theoretically high in these minerals now contain a fraction of what nutritional databases imply. Second, if cortisol is chronically elevated, the demand for pregnenolone is continuously outpacing the dietary supply of the mineral cofactors required to synthesise it. Eating well supports the direction of travel. It does not close the gap when both the precursor diversion and the cofactor depletion are simultaneously active.
What is the connection between low progesterone and perimenopause?
In perimenopause, ovarian progesterone production declines earlier and more steeply than oestrogen, creating a relative oestrogen dominance that intensifies the symptoms described in this post. The adrenal glands become an increasingly important site of progesterone synthesis as ovarian function declines. If those adrenals are simultaneously producing elevated cortisol from chronic stress, pregnenolone is diverted away from adrenal progesterone synthesis as well. The perimenopausal woman under sustained stress experiences both the natural decline of ovarian progesterone and the additional cortisol-driven suppression of adrenal progesterone simultaneously. Significant perimenopausal symptoms warrant GP or gynaecologist assessment for full hormonal evaluation alongside any adaptogenic and mineral support approach.
ONE MORE THING BEFORE YOU GO If you recognise the pattern described in this post but something in your situation does not quite fit the mechanism, leave it in the comments below. We read every comment and respond with what the research says. Tell us your three most persistent symptoms and whether they are cyclically timed. That is enough for us to give you a direct and useful response. If you have already tried dietary changes or supplementation and want to understand why the result was partial rather than complete, that is exactly the kind of question this post was written to answer and we will close it for you specifically. |
Scientific References
- Backstrom, T., Bixo, M., Johansson, M., Nyberg, S., Ossewaarde, L., Ragagnin, G., Savic, I., Stromberg, J., Timby, E., van Broekhoven, F. and van Wingen, G. (2014). Allopregnanolone and mood disorders. Progress in Neurobiology, 113, 88-94.
- Chandrasekhar, K., Kapoor, J. and Anishetty, S. (2012). A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety in adults. Indian Journal of Psychological Medicine, 34(3), 255-262.
- Davis, D.R., Epp, M.D. and Riordan, H.D. (2004). Changes in USDA food composition data for 43 garden crops, 1950 to 1999. Journal of the American College of Nutrition, 23(6), 669-682.
- Epel, E., McEwen, B., Seeman, T., Matthews, K., Castellazzo, G., Brownell, K.D., Bell, J. and Ickovics, J.R. (2000). Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat. Psychosomatic Medicine, 62(5), 623-632.
- Majewska, M.D., Harrison, N.L., Schwartz, R.D., Barker, J.L. and Paul, S.M. (1986). Steroid hormone metabolites are barbiturate-like modulators of the GABA receptor. Science, 232(4753), 1004-1007.
- Netter, A., Hartoma, R. and Nahoul, K. (1981). Effect of zinc administration on plasma testosterone, dihydrotestosterone, and sperm count. Archives of Andrology, 7(1), 69-73.
- Prior, J.C. (2011). Progesterone for symptomatic perimenopause treatment: progesterone politics, physiology and potential for perimenopause. Facts, Views and Vision in ObGyn, 3(2), 109-120.
- Schierbauer, J., Haupt, S., Zimmermann, P., Schumann, U., Zimmer, R.T. and Wachsmuth, N.B. (2023). Magnesium: biochemistry, nutrition, detection, and social impact of diseases linked to its deficiency. Nutrients, 15(4), 1013.
Legal Disclaimer
The information in this post reflects Penantia’s interpretation of available scientific research and is intended for educational purposes only. It does not constitute medical advice, diagnosis, or treatment. If you are experiencing persistent sleep disruption, consult a qualified healthcare provider.
Contents
Subscribe to Our Blog
Never Miss any Updates From Blog!




