You bought the magnesium. You take it every night. You have been taking it for four months. Your sleep is still broken, your muscles still cramp, and you still wake up exhausted. So you search for a better brand. You buy that one. The cycle continues.
The label was accurate the entire time. It genuinely contained 300mg of magnesium per capsule. What the label did not tell you, and what the supplement industry has no legal obligation to disclose prominently, is that the form of magnesium inside that capsule determines how much of that 300mg your body can actually use.
In some cases the answer is 12mg. In others it is 270mg. Same stated dose. A 22-fold difference in what reaches your cells. This is not a fringe claim. It is undergraduate biochemistry. And our research indicates it is the most consistently overlooked variable in the entire supplement market.
BEFORE YOU READ FURTHER This post explains one specific reason why mineral supplements fail to produce the expected results: the form of the mineral determines what percentage of the stated dose reaches your cells. This is a documented biological mechanism and the most significant variable most supplement users have never accounted for. This post is relevant for you if you have been taking a mineral supplement consistently and noticed limited results, or if you are deciding which form to buy. It is not a substitute for clinical guidance if you have a diagnosed mineral deficiency, an absorption disorder such as Crohn’s disease or celiac disease, or if you are taking prescribed mineral supplementation. In those cases the form discussion here is useful context, but your healthcare provider should be guiding the clinical decision. If none of those conditions apply and you are simply trying to understand why your supplement does not seem to be working, read on. |
The Word the Industry Hopes You Never Look Up

Mineral bioavailability is the percentage of a nutrient that is ingested, successfully absorbed through the gut wall, transported to target tissues, and used in a biological function. Not what you swallow. Not what dissolves in your stomach. What completes the entire journey and performs the job it was taken for.
Think of bioavailability like a freight delivery system. A supplier ships 100 packages from a warehouse. Some are lost in transit. Some arrive at the wrong address. Some arrive at the right address but in a damaged form the recipient cannot open. Bioavailability is the percentage of packages that arrive intact, at the correct destination, in a usable condition. A 4 percent bioavailability rate means four packages out of every hundred completed the journey. The other 96 were paid for, processed, and wasted.
Most supplement consumers have never heard this word in the context of what they are buying. Our research indicates this is not accidental. A label that reads ‘300mg Magnesium’ communicates completeness. The bioavailability figure, which would reveal that 288 of those milligrams may pass through the body unused, appears nowhere on the packaging.
DEFINITION MINERAL BIOAVAILABILITY The percentage of a mineral that is ingested, absorbed through the gut wall, transported to target tissues, and used in a biological function. Bioavailability is the true measure of a supplement’s effectiveness, not the milligram count on the label. A 300mg dose of magnesium oxide at 4 percent bioavailability delivers 12mg of usable magnesium to your cells. A 300mg dose of ionic magnesium at 90 percent bioavailability delivers 270mg. Same label. Entirely different biological outcome. |
WHAT TO DO ABOUT IT When evaluating any mineral supplement, the first question is not how many milligrams. It is what form. Find the form listed in the Supplement Facts panel after the mineral name in parentheses. Apply the form hierarchy: oxide and carbonate lowest, citrate and gluconate moderate, glycinate and bisglycinate good, ionic liquid highest. The milligram number is secondary to the form. |
DOES THIS APPLY TO YOU? If you have been taking a mineral supplement for more than four weeks without noticing any of the expected effects, the form is almost certainly the explanation before the dose is. Check the label of what you are currently taking. If it says oxide, carbonate, or sulphate, you are receiving a fraction of the stated dose. If it says glycinate, bisglycinate, malate, or picolinate, the form is good but the dose or timing may need adjustment. If it says ionic or fulvic mineral complex, you are using the highest absorption format available. |
Why the Form of a Mineral Changes Everything
Minerals do not enter cells as raw particles. They enter as ions, single electrically charged atoms that the gut wall and cell membrane recognise and transport through dedicated protein channels. The form a mineral arrives in determines how easily it converts into that ionic state, and therefore how much of it successfully crosses into your bloodstream and then into your cells.
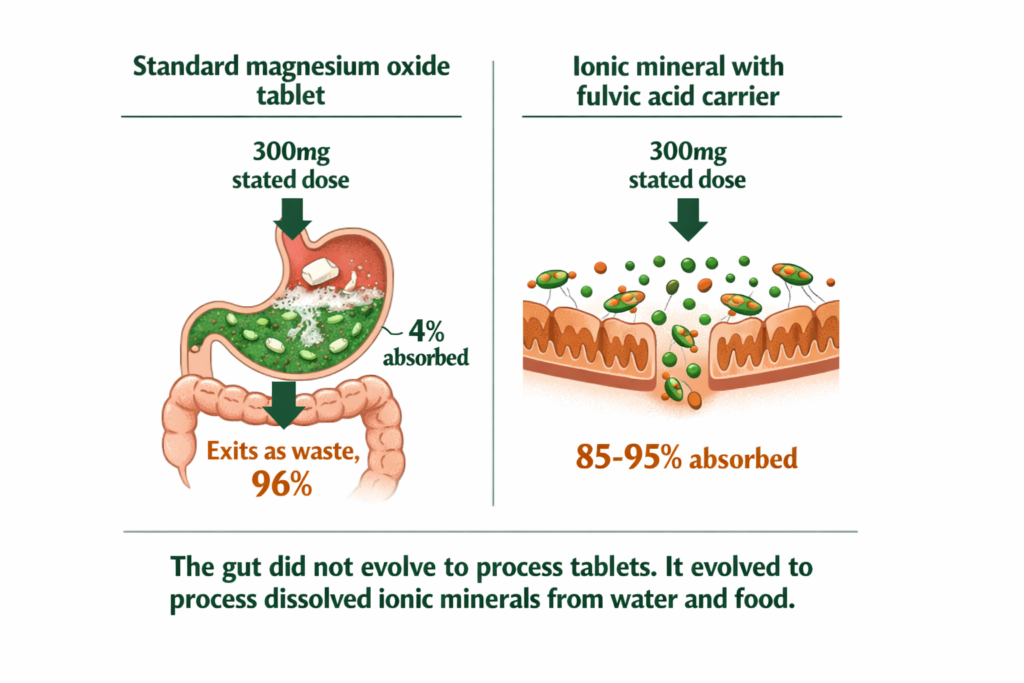
Oxide form: The cheapest and most prevalent form in budget supplements. Magnesium oxide, zinc oxide, and calcium carbonate all fall here. The body must break the mineral-oxygen bond through digestion before any ionic conversion can begin. Research by Firoz and Graber (2001) demonstrated magnesium oxide absorption at approximately 4 percent. A 300mg capsule delivers roughly 12mg of usable magnesium to your cells. The remaining 288mg passes through the digestive tract and exits as waste.
Carbonate and sulphate forms: Slightly more soluble than oxide but still requiring significant digestive conversion. Calcium carbonate, the form in most standard calcium supplements and antacids, has an absorption rate of approximately 22 percent when taken with food, dropping to near zero when taken without it (Heaney et al., 2001). Ferrous sulphate, the most commonly prescribed iron supplement, achieves 10 to 15 percent absorption in individuals with normal iron stores.
Chelated forms: The mineral is bound to an amino acid, which acts as a carrier molecule through the gut wall. Quality chelates such as magnesium glycinate, magnesium malate, and iron bisglycinate achieve 40 to 60 percent absorption. The amino acid bond makes the mineral more recognisable to gut transport proteins and reduces the digestive energy required for absorption. This is why magnesium glycinate is consistently recommended over magnesium oxide for therapeutic use (Walker et al., 2003).
Ionic forms: The mineral is already in its dissolved, electrically charged state. No digestive conversion required. The gut wall recognises it directly. Absorption rates reach 85 to 95 percent. This is the form found in natural mineral-rich spring water that has traveled through geological rock formations over centuries, and it is the form found in naturally occurring Shilajit resin, where a full spectrum of ionic minerals is delivered alongside fulvic acid, a small organic molecule that actively transports minerals across both the gut wall and the cell membrane.
WHAT TO DO ABOUT IT Moving from oxide to chelated or ionic forms of the same mineral is the single most reliable upgrade available to any supplement user. No increase in dose required. No new products required in most cases. Just a different form of what you are already taking, delivering a fundamentally different biological outcome. |
DOES THIS APPLY TO YOU? If you have tried switching from oxide to glycinate and noticed a clear difference within two to three weeks, you were receiving six to twenty times more usable magnesium from the same stated dose and the difference was real, not placebo. If you switched to glycinate and still noticed limited results, the gut delivery step may be the remaining variable: ionic forms via fulvic acid cross both the gut wall and the cell membrane more efficiently than chelated forms alone. If you have a diagnosed gut condition such as Crohn’s, celiac, or low stomach acid, dissolution-dependent formats including tablets and capsules underperform further and liquid or ionic formats are significantly more appropriate. |
Three Minerals. The Same Principle Applied.
The bioavailability gap is not a magnesium-specific problem. It applies uniformly across the mineral spectrum. Three examples demonstrate the scale of the issue.
Iron: The Vitamin C Cofactor Most People Miss
Iron exists in two dietary forms: haem iron from animal sources, and non-haem iron from plant sources and most supplements. Non-haem iron, the form in the majority of iron tablets sold globally, has a base absorption rate of 2 to 20 percent depending on gut conditions (Hallberg et al., 1989). That range is not an error. It reflects how dramatically absorption of iron is affected by what you consume alongside it.
Vitamin C is the most significant absorption cofactor for non-haem iron known to nutritional science. Research confirms that consuming 200mg of vitamin C alongside non-haem iron increases absorption by 2.9 times in individuals with normal iron stores, and up to 6 times in individuals with iron deficiency (Lynch and Cook, 1980). The mechanism is direct: vitamin C reduces ferric iron to ferrous iron, the form gut transport proteins actually recognise. Without that conversion, a significant proportion of the iron in your tablet passes through without being absorbed.
Most iron supplements are taken without vitamin C. Most iron-deficient individuals are never told this. The supplement is not failing them. The instruction to take it with orange juice or a vitamin C tablet was simply never included on the label.
A parallel cofactor relationship exists for calcium. Calcium requires vitamin D for active transport across the gut wall. Without adequate vitamin D, calcium absorption drops from approximately 30 to 40 percent to as low as 10 to 15 percent regardless of the form taken (Heaney, 2008). Calcium supplement absorption figures in research literature assume adequate vitamin D status. In populations with widespread vitamin D deficiency, the real-world absorption rates are considerably lower than any label implies.
WHAT TO DO ABOUT IT For iron: take non-haem iron supplements with 200mg of vitamin C. Orange juice, a vitamin C tablet, or a capsule form containing both. This single change can increase iron absorption by up to six times in deficient individuals without changing the dose. For calcium: check vitamin D status before attributing poor calcium supplementation results to the supplement. Calcium cannot perform its absorption function without adequate vitamin D regardless of the form or dose taken. |
DOES THIS APPLY TO YOU? If you have been supplementing iron for more than eight weeks without meaningful improvement in energy or ferritin levels, and you have not been taking it with vitamin C, that is the most likely explanation before dose or form. If you have been taking vitamin C alongside it and still noticing limited results, haem iron from animal sources or iron bisglycinate in a chelated form is the next step. If iron deficiency was diagnosed by a blood test and is not responding to supplementation, consult your GP as absorption disorders warrant clinical investigation rather than form changes alone. |
Magnesium: The Oxide vs Glycinate vs Ionic Comparison
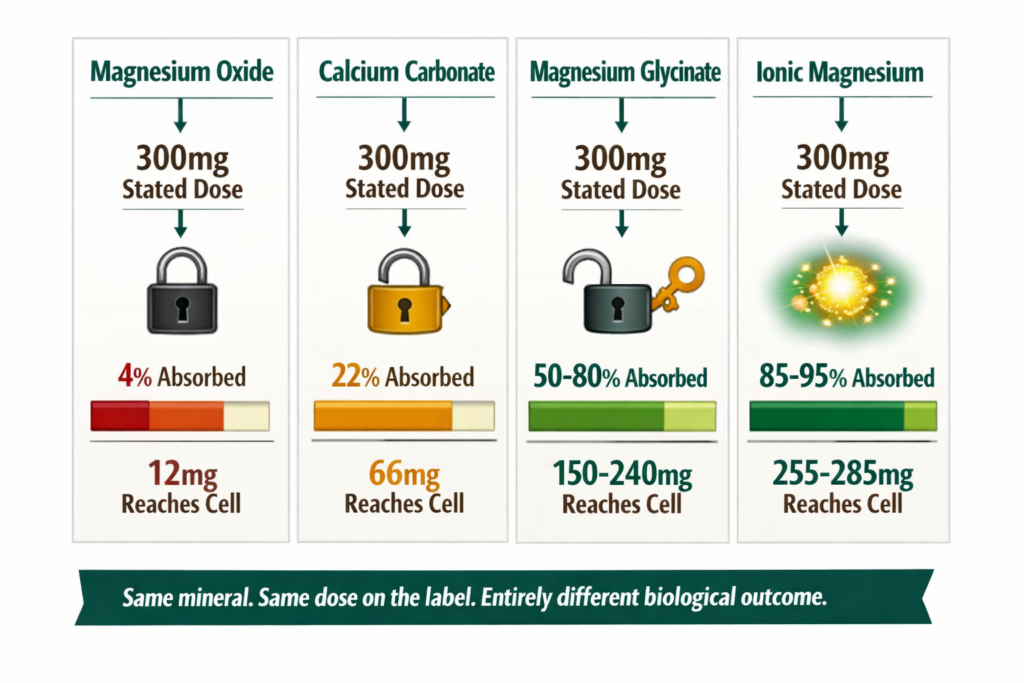
The magnesium market is where the bioavailability gap is most visible and most commercially exploited. The following comparison uses the same stated dose of 300mg across all forms.
Form | Absorption Rate | From 300mg, cells receive | Market Position |
|---|---|---|---|
Magnesium Oxide | 4% | ~12mg | Dominant, cheapest to produce |
Magnesium Citrate | 25-30% | ~75-90mg | Common, mid-tier pricing |
Magnesium Glycinate | 50-80% | ~150-240mg | Premium, amino acid chelated |
Ionic Magnesium in Water | 85-95% | ~255-285mg | Rare, highest bioavailability |
The magnesium citrate vs oxide question is the most commonly searched comparison in this space. Someone who switched from oxide to citrate and noticed a difference was not experiencing placebo effect. They were receiving six times more usable magnesium from the same stated dose. The magnesium oxide vs glycinate comparison produces an even starker outcome: a person taking glycinate receives up to 20 times more usable magnesium from the same stated dose of oxide (Schuette et al., 1994). The label for both products reads identically in milligrams. The biological outcome is not remotely comparable.
WHAT TO DO ABOUT IT If you are currently taking magnesium oxide and experiencing sleep disruption, muscle cramps, anxiety, or fatigue that should be responding to magnesium: switch to magnesium glycinate or ionic magnesium before concluding supplementation does not work for you. The form change alone, at the same stated dose, is the most significant intervention available. Do not increase the milligrams. Change the form. |
DOES THIS APPLY TO YOU? If you switched to magnesium glycinate from oxide and noticed clear improvement within two weeks, you confirmed the form was the variable. If you have been taking magnesium glycinate consistently for six or more weeks at a meaningful dose and symptoms are not responding, the cortisol depletion pathway may be consuming magnesium faster than the supplement is replacing it. Both the form and the rate of depletion matter simultaneously. If you are under sustained stress, both mineral restoration and adaptogenic cortisol support are relevant. |
Zinc: The Picolinate and Bisglycinate Advantage
Zinc follows the same pattern. Zinc oxide, used in many multivitamins because of its low cost, has a bioavailability of approximately 49 to 61 percent under optimal conditions, dropping significantly in the presence of dietary phytates found in grains and legumes (Sandstead, 1991). Zinc picolinate and zinc bisglycinate, both chelated forms, achieve 60 to 80 percent absorption with far less sensitivity to dietary interference. Zinc gluconate, the form in most lozenges, sits in the middle at 60 to 70 percent.
The zinc absorption rate differences across forms are smaller than magnesium, but the cofactor sensitivity adds a layer most consumers are unaware of: phytic acid in wholegrains, coffee, and tea significantly inhibits zinc absorption regardless of the form taken. A person taking zinc with their morning coffee and oat porridge is absorbing a fraction of what the label implies, regardless of whether the form is good.
WHAT TO DO ABOUT IT Take zinc on an empty stomach or with a small protein-containing meal for highest absorption. Avoid taking it alongside coffee, tea, or a high-grain breakfast. For form: picolinate or bisglycinate over oxide or gluconate. For immune function specifically, the intracellular delivery step matters beyond gut absorption: ionic zinc via fulvic acid crosses both the gut wall and the cell membrane, reaching the T-cells where zinc does its immune work. |
DOES THIS APPLY TO YOU? If you are taking zinc oxide in a multivitamin alongside your morning coffee and oat porridge, you are likely absorbing a very small fraction of the stated dose due to both form limitations and cofactor competition. Separating zinc from coffee and grains by at least two hours, and switching to picolinate or bisglycinate form, addresses both variables. If zinc supplementation at good form and good timing is still not producing the expected results for immunity or testosterone, the cortisol depletion rate is the next variable to address alongside the mineral supply. |
Standard Thinking vs. The Biological Reality
Standard Thinking | The Biological Reality |
|---|---|
300mg on the label means 300mg in your body | 300mg on the label means 12mg to 285mg in your cells depending on the form. The label states the dose. It does not state the bioavailability. |
All magnesium supplements are equivalent | Magnesium oxide and magnesium glycinate produce a 20-fold difference in cellular delivery from the same stated dose. They are not equivalent in any biologically meaningful sense. |
Iron supplements work if you take them consistently | Iron supplements work if you take them with vitamin C consistently. Non-haem iron absorption can increase by up to six times with 200mg of vitamin C alongside each dose. |
Approved and sold means it works | Approved means it is safe. Regulatory bodies assess safety, not efficacy. A magnesium oxide supplement at 4 percent absorption is completely safe and meets every regulatory standard. It may not correct a magnesium deficiency. |
More milligrams means more benefit | More milligrams of the wrong form means more expensive waste. The form hierarchy, not the milligram count, determines biological outcome. |
Natural food sources are nutritionally complete | Soil mineral depletion since 1950 has reduced mineral content of food by 40 to 80 percent. Eating well is necessary. It is no longer sufficient as a sole mineral strategy. |
The Regulatory Gap Nobody Explains to You
Regulatory bodies including the Therapeutic Goods Administration in Australia, the Food Safety and Standards Authority of India, and the Food and Drug Administration in the United States assess supplements through a safety framework. The central question they answer is: will this product harm the person who takes it?
Safety is regulated. Efficacy is not.
A magnesium oxide supplement at 4 percent absorption is completely safe. It passes through the body, exits harmlessly, and causes no toxicity. It therefore meets every regulatory standard it was assessed against. The regulatory body has done its job correctly. It was never asked to assess whether the product delivers the biological benefit the consumer assumes it provides.
This is not corruption. It is a structural gap between what supplement regulation was designed to do and what consumers believe it does. The consumer assumes that if a product is approved, labelled accurately, and sold legally, it works as implied. The regulatory framework was never designed to guarantee that. It was designed to guarantee the product does not cause harm.
The result is a legal market where a product can contain precisely what the label states, carry full regulatory approval, and deliver a fraction of its implied biological benefit without violating a single rule. Understanding this gap is the single most valuable piece of consumer knowledge in the supplement industry. It reframes every purchase decision you will ever make.
WHAT TO DO ABOUT IT Apply the same scepticism to the phrase ‘clinically studied’ on supplement packaging that you now apply to milligram counts. The question is always: studied in what form, at what dose, in what population, and against what outcome measure? A study showing benefit from magnesium glycinate does not validate a product containing magnesium oxide. The study applies to the form studied, not to all products with the same mineral name on the label. |
DOES THIS APPLY TO YOU? If you have concluded that supplements do not work for you after trying them consistently and noticing nothing, your conclusion may be accurate for the specific form you tried. It is not accurate as a general statement about mineral supplementation. The regulatory gap means the products most likely to underperform are also the most widely available and most affordable. The products most likely to work are typically more expensive and less prominently marketed. This is not a coincidence. It is the predictable outcome of a market where efficacy is unregulated. |
How Soil Depletion Compounds the Problem
The bioavailability crisis is not limited to supplements. It extends to the food that was supposed to provide minerals naturally.
Research published in the Journal of the American College of Nutrition analysed United States Department of Agriculture nutritional data for 43 common garden crops and found reliable declines in protein, calcium, phosphorus, iron, riboflavin, and vitamin C between 1950 and 1999 (Davis et al., 2004). The researchers attributed the declines primarily to agricultural practices that prioritise yield and growth rate over mineral density. A larger, faster-growing vegetable draws from the same soil mineral pool but distributes those minerals across greater mass. The mineral content per gram drops.
This creates a compound problem. The food delivers less than it once did. The supplement meant to compensate delivers a fraction of what the label implies. The person taking that supplement with coffee or a calcium-rich meal compounds the absorption further through cofactor competition. The cumulative deficit is substantial, invisible on standard blood panels, and experienced as the chronic low-grade symptoms that bring people to posts like this one.
WHAT TO DO ABOUT IT The soil depletion context changes the calculus from ‘do I need a supplement’ to ‘which supplement will actually deliver what the depleted food supply can no longer provide.’ Dietary improvements are necessary and valuable. They are not sufficient as a sole mineral strategy in a food system operating on mineral-depleted soil. The form hierarchy matters more in this context, not less. |
What Natural Ionic Mineral Sources Do Differently
The distinction between oxide, chelated, and ionic mineral forms points toward a consistent conclusion: the closer a mineral source is to the ionic state found in nature, the higher the bioavailability and the lower the dependence on digestive conversion.
Naturally occurring Shilajit resin, formed over geological timescales through the compression and decomposition of mineral-rich plant matter in high-altitude rock formations, delivers a full spectrum of ionic minerals in their naturally dissolved state. This is not a marketing claim. It is a description of the geological process that produces the compound. The fulvic acid present in authentic Shilajit serves as a transport molecule, actively carrying ionic minerals across the gut wall and through cell membranes, the same transport mechanism described in the cellular hydration literature (Bhattacharyya et al., 2009).
The significance of this is not that Shilajit is a miracle product. It is that the delivery mechanism, ionic minerals plus a fulvic acid transport carrier, represents the highest bioavailability pathway available for mineral absorption. It is the form the human gut evolved alongside. No digestive conversion required. No cofactor dependency. No form-related absorption ceiling.
The full cellular mechanism behind why ionic mineral delivery produces different outcomes from standard supplements is covered in our post on why water stops entering your cells and what the mineral supply has to do with it.
How to Read a Supplement Label Properly
Most people can read a supplement label. Very few people know what to look for beyond the milligram number. Here is the functional reading framework.
Step one: Find the form, not just the mineral name. After the mineral name in the Supplement Facts panel you will find the compound name in parentheses. ‘Magnesium (as Magnesium Oxide)’ tells you the form. ‘Magnesium (as Magnesium Bisglycinate)’ tells you a different form entirely. If the form is not stated, it is almost always the cheapest option.
Step two: Apply the form hierarchy. Oxide and carbonate forms: lowest bioavailability. Citrate, gluconate, and sulphate forms: moderate. Glycinate, malate, bisglycinate, and picolinate forms: good. Ionic liquid forms: highest. This hierarchy applies to magnesium, calcium, zinc, iron, and most other minerals.
Step three: Check the cofactor requirements. Iron requires vitamin C. Calcium requires vitamin D. Magnesium and zinc compete for the same gut transporters when taken together in high doses. If the label does not address cofactors, the manufacturer is not accounting for real-world absorption conditions.
Step four: Consider the delivery format. Tablets require dissolution before absorption begins. Capsules dissolve faster. Liquids and ionic solutions require no dissolution and absorption begins immediately. If the gut environment is compromised through low stomach acid, stress, or inflammation, dissolution-dependent formats underperform further.
WHAT TO DO ABOUT IT This four-step framework takes 60 seconds to apply to any supplement label. Step one eliminates most budget products immediately. Step two separates good forms from acceptable forms. Step three tells you what you need to take alongside it. Step four tells you whether the delivery format matches your gut capacity. A product that passes all four steps is worth buying. A product that fails step one is not worth buying regardless of how good the marketing is. |
DOES THIS APPLY TO YOU? If you apply this framework to what you are currently taking and discover the form is oxide or carbonate, the decision is straightforward: switch form before increasing dose or abandoning the mineral entirely. If the form is already good but results are still limited, step three and step four are the variables to check. If all four steps check out and results are still limited after six or more weeks, depletion from another source such as chronic stress or dietary phytate competition is the remaining variable. |
The Objection Worth Addressing
If the absorption rates are this different, surely doctors and pharmacists would be recommending the higher bioavailability forms as standard.
In clinical medicine, they often do. Magnesium glycinate and magnesium malate are commonly recommended by integrative practitioners over magnesium oxide. Iron bisglycinate is prescribed in preference to ferrous sulphate for patients who experience gastrointestinal side effects. Calcium citrate is recommended over calcium carbonate for patients with low stomach acid.
The gap is not in clinical practice at the specialist level. It is in the mass consumer supplement market, where products are purchased without clinical guidance, labels communicate milligrams but not bioavailability, and price signals quality inversely. Oxide forms are cheaper, so they appear more affordable and therefore more accessible. The consumer who cannot afford the glycinate form receives a product that is almost biologically inert at the same stated dose.
The second layer of this objection is the assumption that because something has been taken for decades without obvious harm, it must be working. Magnesium oxide at 4 percent absorption does not cause harm. It also does not correct magnesium deficiency in any meaningful timeframe at standard doses. The absence of adverse effects has been consistently misread as evidence of efficacy. It is evidence only of safety, which is the regulatory standard the product was assessed against, and nothing more.
Frequently Asked Questions
Why am I not absorbing magnesium despite taking it daily?
The most common reason is form. If your supplement contains magnesium oxide, you are absorbing approximately 4 percent of the stated dose. Switching to magnesium glycinate or magnesium bisglycinate immediately increases absorption to 50 to 80 percent from the same stated milligrams. The second reason is timing: magnesium taken with calcium in high doses competes for the same gut transporters, reducing absorption of both. Take them separately where possible. The third reason, for people who have already switched to glycinate and still notice nothing, is that the cortisol depletion pathway is consuming magnesium faster than the supplement is replacing it. Both the form and the depletion rate need to be addressed.
Why do vitamins and supplements not work for some people?
Beyond form and cofactor issues, three factors consistently reduce supplement efficacy: low stomach acid, which reduces the ability to dissolve and ionise minerals from tablet forms; gut inflammation or permeability issues, which impair transport protein function; and the rate of mineral depletion from sources such as chronic stress and dietary phytate competition exceeding the rate of supplementation. These factors explain why two people taking identical supplements can have measurably different outcomes without any difference in the product itself.
What is the best calcium supplement for absorption?
Calcium citrate consistently outperforms calcium carbonate in clinical absorption studies, particularly in individuals over 50 where stomach acid production declines naturally (Heaney et al., 2001). Calcium citrate malate achieves the highest absorption rate among tablet forms. Calcium from food sources and ionic mineral water is absorbed via different pathways and is less sensitive to stomach acid availability. Vitamin D status remains the most significant variable: no calcium supplement performs well in vitamin D deficient individuals regardless of form.
Does zinc absorption rate vary significantly between supplement forms?
Yes. Zinc picolinate and zinc bisglycinate consistently outperform zinc oxide and zinc gluconate in head-to-head absorption studies. Zinc absorption is also significantly impaired by phytic acid in wholegrains and legumes, and by calcium in high doses. Taking zinc on an empty stomach or with a small protein-containing meal achieves the highest absorption rate for any form. For immune function specifically, where zinc needs to reach T-cells inside immune cells rather than just the bloodstream, ionic zinc via fulvic acid is the most complete delivery format available.
ONE MORE THING BEFORE YOU GO Are you currently taking a mineral supplement? Tell us the form listed on your label in the comments. We will tell you your real-world absorption rate and whether a form change is worth considering. If you have already switched forms and noticed a result, tell us that too. What changed, when it changed, and at what dose. That information is useful to other readers and we will respond to every comment with the research behind it. |
The Conclusion That Changes Every Purchase You Make
You have been measuring your supplement intake in milligrams. The number that actually governs your biological outcome is the percentage of those milligrams that completes the full journey from the capsule to the inside of your cells.
That percentage is determined by form, by cofactors, by delivery format, and by gut condition. None of these variables appear prominently on any supplement label. All of them are knowable. And knowing them converts supplement literacy from reading a label into actually understanding what you are purchasing and why.
Our research indicates the single most reliable upgrade available to any supplement user is to move from oxide and carbonate forms to chelated or ionic forms of the same mineral. No increase in dose required. No new products required in most cases. Just a different form of what you are already taking, delivering a fundamentally different biological outcome.
The label will still read the same milligrams. What happens inside your body will not be the same at all.
Scientific References
- Bhattacharyya, S., Pal, D., Gupta, A.K., Ganguly, P., Majumder, U. and Bhattacharya, S.K. (2009). Beneficial effect of processed Shilajit on swimming exercise induced impaired energy status of mice. Pharmacologyonline, 1, 817-825.
- Davis, D.R., Epp, M.D. and Riordan, H.D. (2004). Changes in USDA food composition data for 43 garden crops, 1950 to 1999. Journal of the American College of Nutrition, 23(6), 669-682.
- Firoz, M. and Graber, M. (2001). Bioavailability of US commercial magnesium preparations. Magnesium Research, 14(4), 257-262.
- Hallberg, L., Brune, M. and Rossander, L. (1989). Iron absorption in man: ascorbic acid and dose-dependent inhibition by phytate. American Journal of Clinical Nutrition, 49(1), 140-144.
- Heaney, R.P. (2008). Vitamin D and calcium interactions: functional outcomes. American Journal of Clinical Nutrition, 88(2), 541-544.
- Heaney, R.P., Dowell, M.S. and Barger-Lux, M.J. (2001). Absorption of calcium as the carbonate and citrate salts, with some observations on method. Osteoporosis International, 11(4), 401-406.
- Lynch, S.R. and Cook, J.D. (1980). Interaction of vitamin C and iron. Annals of the New York Academy of Sciences, 355, 32-44.
- Sandstead, H.H. (1991). Zinc bioavailability: a critical examination. American Journal of Clinical Nutrition, 53(2), 403-408.
- Schuette, S.A., Lashner, B.A. and Janghorbani, M. (1994). Bioavailability of magnesium diglycinate vs magnesium oxide in patients with ileal resection. Journal of Parenteral and Enteral Nutrition, 18(5), 430-435.
- Walker, A.F., Marakis, G., Christie, S. and Byng, M. (2003). Mg citrate found more bioavailable than other Mg preparations in a randomised, double-blind study. Magnesium Research, 16(3), 183-191.
Legal Disclaimer
The information in this post reflects Penantia’s interpretation of available scientific research and is intended for educational purposes only. It does not constitute medical advice, diagnosis, or treatment. If you are managing a diagnosed mineral deficiency or taking prescribed mineral supplementation, consult your healthcare provider before making changes.
Contents
Subscribe to Our Blog
Never Miss any Updates From Blog!
